My laboratory recently published some results of our ongoing research project designed to identify novel human-specific genes in the human genome. This work began with what we thought was a very naive and probably fruitless approach: we aligned human chromosomes with their counterparts from the other apes and looked for regions that aligned poorly and were thus unique to humans. Our plan was to begin with the smallest chromosome – chromosome #21 – then work our way toward the largest ones. We had hoped that eventually we might find something worth pursuing that others had overlooked, but we figured it would take months, maybe years. But during the second week of this work…
To our great surprise, we found that the small arm of human chromosome #21 did not align well to its equivalent in any of the other apes. However, among the other apes, the entire chromosome aligned very well with each other. This means that humans are the outliers and that something strange has occurred in the short arm of our chromosome #21. Here is a crude picture of the alignment results:

As we zoomed in closer, we noticed two things: a) the entire short arm of human chromosome #21 is kind of just a mess, and b) a 1.2 million base-pair region has undergone extensive genomic rearrangements not observed in the other apes. As we continued our analysis, we noticed that this region of the chromosome is home to 8 different microRNA genes, grouped into three families, and that none of these genes are found in the other apes (although there are similar DNA sequences).

Most interesting of all, these microRNA genes are nestled within an array of ribosomal RNA genes, which led us to our hypothesis that these microRNA genes were created through repeated rounds of segmental duplication. Ribosomal RNAs have a very complex shape, with lots of “hairpin regions” of secondary structure (like microRNAs have), and the genes for these RNAs exist in large arrays that are transcribed and then extensively processed. It’s conceivable that a small chunk of an rRNA gene could form a nascent microRNA gene, and that is our hypothesis for the origin of these genes.
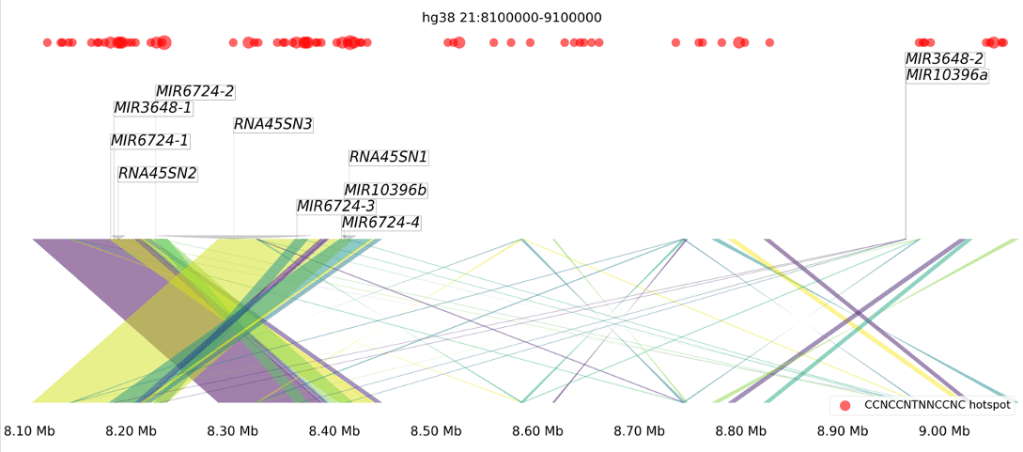
To our knowledge, this is the first evidence that microRNA genes can be created de novo through genomic rearrangements of ribosomal genes. In addition, we performed target gene prediction for these microRNA and found an abundance of genes related to embryonic development of the nervous system among the targets. This leads to the fascinating possibility that microRNA genes may have played an under-appreciated role in the unique evolutionary trajectory of humans. My lab is now fully focused on microRNA gene evolution in humans, Neanderthals, and Denisovans, so look out for updates about that later this year.
You can read the full paper here. And a press release about the paper, featuring how this work was done mostly by undergraduate students at John Jay College. I am enormously proud of my mentees, and all four of them are at the beginning of what I know will be long and successful scientific careers.
-NHL







Leave a comment